- Joined
- Sep 12, 2007
- Messages
- 9,399
- Points
- 113
Copper also has a higher heat capacity than aluminum, meaning it takes more energy to raise the temperature of one kg of copper 1 degree than it does for aluminum.
But copper is more dense, and therefore has more mass per unit volume. Our applications are generally limited by volume, and the heat capacity is actually about the same per unit volume.
The thermal conductivity of most aluminum alloys is around 100-130W/mK
You mean to say an alloy of aluminum is half as conductive as pure aluminum? That's a rather large difference for such a small change in material. Source?
copper will distribute the heat evenly through the heatsink 4 times faster than an aluminum heatsink.
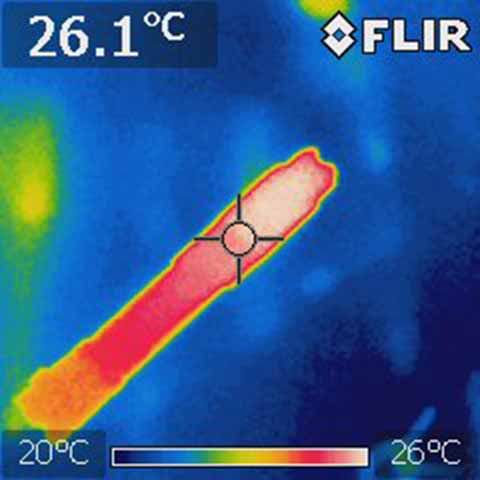
That's a bit misleading since the heat isn't created all at once. If you actually take the temperature of various parts of aluminum, it's virtually all the same. Take this image of the arctic for example. The junction of the tail cap provides FAR more resistance than even several inches of aluminum. If it were made of copper, the image would not be much different
Last edited:





